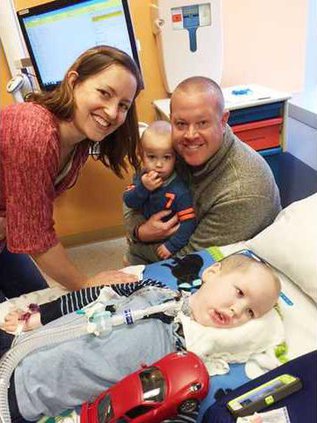
The seven years of young Charleston “Cash” Goeppert’s life have been marked by a lot of nevers — never taken his first step, never crawled, never sat up on his own — but now, thanks to unwavering advocacy from his parents and a new drug treatment, Cash is starting to have a list of firsts.
“We are getting to experience our son in a way we never got to do,” said Cash’s father Cameron Goeppert. “It’s like he’s an infant again, like we’ve hit a restart.”
Cash was born with spinal muscular atrophy, which is a neuromuscular disease that causes muscle atrophy and weakness. It’s caused by a defect in the Survival Motor Neuron 1 gene, which obstructs the growth of proteins that feed and develop muscles. As a result of SMA, Cash’s muscle movements are drastically limited. He is dependent on a ventilator to breath and a feeding tube for his nutrients. He utilizes various technology to communicate and a special power chair for mobility. Cash has a more severe form of SMA, classified as Type 1. The onset occurs in the first few months of an infant’s life and the typical life span is around two to three years.
At 7-years-old, Cash has already defied the odds, and now with the new drug treatment Spinraza, he is helping blaze a path for other children with SMA. Spinraza is the first drug in the United States that has been approved by the Food and Drug Administration to treat SMA. A teaspoon size dose of the drug is injected into the spinal fluid and goes to work on the back-up gene that produces protein. Cash is getting four “loading up” doses of the treatment and after that he will need a maintenance shot every four months. Cash is getting his treatment at Sutter Children’s Center in Sacramento, which is the first facility in the region to offer the treatment.
“Sutter has been very forward thinking on this and wanted to tackle the challenge,” Cameron said. “Sutter will be the example for all others. They are ready to slay the dragon.”
So far, Cash has received two of his four initial treatments, and the improvements have been quick-coming. After his first treatment he was able to move all his fingers and purposeful movement of his jaw. His medical providers could also measure that he was breathing deeper and taking breaths over the ventilator.
“I’m watching my son reinvent himself because of a medicine,” Cameron said. “I’m watching God’s work.”
Dr. Paul Chretien and Dr. Shailesh Asaikar are the pediatric neurologists who are treating Cash with the Spinraza at Sutter. After watching patients die from the disease over three decades, Chretien got emotional when asked to describe the improvements he was seeing with the new drug.
“We are giving hope to the hopeless,” Chretien said. “We are now able to intervene and help and not just watch as our patients go slowly down.”
For Cash, Chretien hopes to one day see him reach some milestones that previously had been out of reach.
“I hope one day he can feed himself, hold his mother’s hand, smile and blow bubbles,” Chretien said.
Getting Cash the treatment took some major advocating on the part of his parents. The maker of Spinraza is Biogen and Ashley Goeppert said she had their webpage on a constant refresh on Dec. 23 when it was supposed to be approved.
“I kept hitting refresh all day long and when it finally came up that it was available, it was like the best Christmas present ever,” Ashley said. “I knew there were going to be a lot of steps to go through to get the drug and I didn’t want to sit around a wait, so I texted Cash’s physician and we put things in motion.”
The treatment has a hefty price tag, though. The doses are $125,000 each and while the first four were approved by the family's insurance, they have no idea yet what the co-pay might be or what the maintenance shots will cost them.
And while Ashley and Cameron would be delighted to see their son reach some of the typical milestones of growing up, they are absolutely overjoyed at the possibilities for his future.